 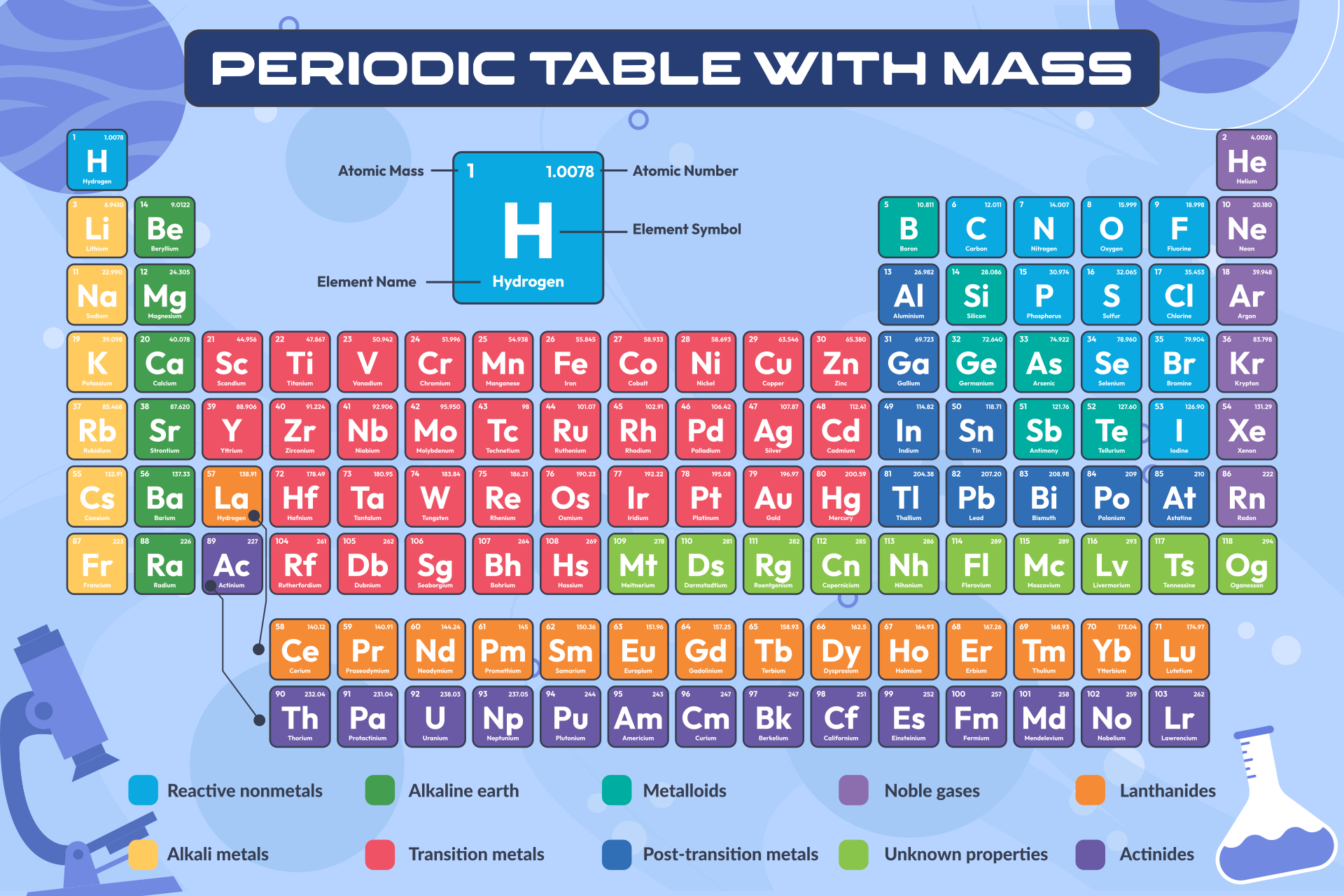
The lessons learned in this post will continue through the study of chemical reactions and equations. We also learned how to calculate the molar mass of a compound using the periodic table and how to convert mass into moles. We learned about Avogadro’s number and how it relates to the mole, which is a unit used to express the amount of a substance. In conclusion, understanding molar mass is an essential concept in chemistry as it allows us to relate the amount of a substance to the number of particles present in it. Periodic Table: Unique 4-letter Combinations, Part IV. For a fully interactive version with orbitals, isotopes, compounds, and free printouts, visit. Specifically, the number is defined as 6.022 \times 10^. Next, using the periodic table, find the atomic mass in g/mol of each element (the molar mass of an element is equal to its atomic mass): Molar Mass (g/mol) e (Electron) 0.000548579909: We use 5.4857990888×10-4 for the electron molar mass. Molar masses of Elements Can you name the Molar masses of Elements By chemistrymaster. Design Copyright © 2017 Michael Dayah (). 
Example 3: Finding the Mass of a Number of MoleculesĪvogadro’s number is a fundamental constant that represents the number of particles (atoms, molecules, ions) in one mole of a substance.How to Find Molar Mass Using the Periodic Table.The Mole and its Relationship to Avogadro’s Number.We have excluded elements above 108 on this periodic table for the simple reason that the larger elements don’t really exist in the real world. Learn more about the elements with fully descriptive write-ups and visualizations. You might find a table of elements showing element 115 or even 118 elements total. Discover the interactive periodic table of elements with Ptable, a web-based tool that lets you explore the properties, trends, orbitals, isotopes, and compounds of each element. No Internet needed - applies only to the smartphone app. The PDF is suitable for high-resolution color printing for desk or wall-chart display. Elemental comparison - compare elements values right next to each other. The periodic table contains NIST’s latest critically evaluated data for atomic properties of the elements. The web app features a completely overhauled molar mass caluclator. There is a Netflix documentary about this topic entitled Bob Lazar: Area 51 & Flying Saucers. Molar mass calculator: Simple entry field for chemical formulas. The elements above 92 are strange, like for example element 115 that was reportedly found at Area 51. Each element square contains all 118 of elements with the element number, symbol, name, atomic mass, and most common oxidation number. Reviewing the way of finding the charge on an atom may help, too. The parenthesis are a scientific way of saying this number is guesstimated, at best, and we really don’t know much about these elements because they are artificial and tend to disintegrate rapidly. This periodic table with charges is a useful way to keep track of the most common oxidation numbers for each element. You might note in the periodic table the mass numbers are in parenthesis for elements 93 and above. At least you won’t be finding any in this lifetime. As such, elements above 92 don’t really exist, except under special laboratory conditions. All elements above atomic number 92 are created artificially in laboratories, and generally they are extremely unstable and tend to disintegrate rapidly. As such, it’s highly unstable and unable to exist for very long. Divide the molar mass by the Empirical Formula Mass. It is element 94, making it bigger than uranium. Look at periodic table or periodic chart to find the masses of each element. 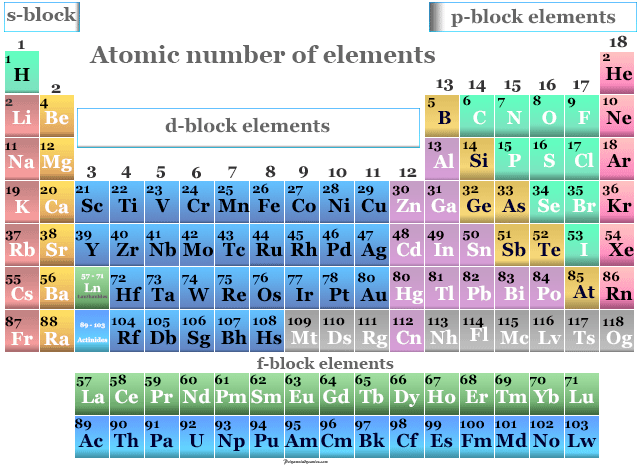
In 1940, a new element, plutonium, was created in a laboratory. Element 92 is uranium, the biggest element that occurs naturally on Earth. Before the year 1940, it was believed that only 92 elements existed. (See the original work of Dmitri Mendeleev here.) At that time, scientists were still discovering new elements almost every year. The original table of the elements, credited to Dmitri Mendeleev in 1871, had only 56 elements. The table of elements shown here has 108 elements. 
0 Comments
Leave a Reply.AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
